
g.Estim PRO is a constant current, bi-phasic electrical stimulator intended to stimulate neural tissue during functional brain mapping procedures prior to cortical resections in the vicinity of essential cortex. The device is CE certified (Europe) and FDA-cleared (USA) for use in human patients for investigations like electrical cortical stimulation (ECS) mapping. Therefore, g.Estim PRO must be used by medically trained and qualified personnel within a medical environment.
g.Estim PRO has an applied part of type BF with connectors for bipolar stimulation electrodes (anode and cathode). The device is controlled by a computer via an USB connection. It also has digital outputs for synchronization with other devices.
g.Estim PRO includes an impedance check and measures the actually applied stimulation current and voltage for verification purposes. With its 80V compliance voltage, it is perfectly suited for use with standard as well as high-impedance electrodes, and is able to produce the required stimulation current. For closed-loop experiments, a g.USBamp or g.HIamp is used with g.HIsys to record EEG/ECoG data, perform real-time analysis and control the g.Estim PRO for cortical stimulation (for research purposes; requires the g.Estim PRO research software upgrade).
g.ESTIM PRO is available with an application programming interface (API) that allows to tune stimulation parameters on the fly with g.HIsys and a g.tec biosignal amplifier. Users are able to record from a brain area and to extract parameters and based on the result drive accordingly the brain stimulation.
| Delivers bi-phasic constant current pulses |
| Application programming interface (API) to configure stimulation parameters on the fly |
| Stand-alone device that can be controlled in real-time from a computer system |
| Can send triggers to other devices for synchronization |
| Includes electrode impedance check and stimulation current monitoring |
| CE-certified and FDA-cleared medical device for use in human patients |
| Stimulus current output | ± 0.2 – 15 mA (±10% or 50µA whichever greater) |
| Phase shape | rectangular |
| Phase duration | 0.1 - 1.0 ms in 10 µs increments (± 10% or ± 20 µs whichever greater) |
| Pulse rate | 2 – 100 pulses/second in 0.1 increments (±10%) (Pulse onset interval from 500 ms down to 10 ms) |
| Train duration | 1 pulse – 20 seconds |
| Power supply | 2 x 9 V battery, USB – connection |
| Certification and Standards | IEC 60601-1, IEC 60601-1-2, IEC 60601-2-40, IEC 62304, IEC 62366, ISO 14971 |
The hand switch is an accessory for the g.Estim PRO. Users connect the hand switch to the hand/foot switch input of the g.Estim PRO. With the hand switch, you can start stimulation by pressing the blue STIMULATE button after the device has been set “active” via the software and the “active” LED is on. During stimulation, the hand switch allows users to abort stimulation by pressing the grey ABORT button.
The foot switch is another accessory for the g.Estim PRO that can be connected to the hand/foot switch input of the g.Estim PRO. When the foot switch is used and pressed, the stimulator accepts a cue to start stimulation from the software or from devices connected to the digital input ports. When the foot switch is released, stimulation is aborted.
The Switching Unit for g.Estim PRO allows to select certain electrodes that should deliver the electrical stimulation. The switching unit is available with 64, 128 or 256 channels and is connected to the g.HIamp to measure EEG or ECoG data. A control interface allows to select certain electrode channels that should deliver the electrical stimulation pulses from g.Estim PRO. When g.HIsys is used, the amplifier block can read in the EEG or ECoG data and analyze it.
g.tec provides an intracranial stimulator with 80V compliance voltage for electrical stimulation of the cortex with ECoG grids or strips or for depth electrodes. This can be used e.g. to stimulate the sensory cortex to add a touch feeling if robotic devices are controlled with the BCI or for deep brain-stimulation experiments e.g. with Parkinson patients. Stimulation parameters such as on-set time or stimulation current can be set and triggered in real-time from Simulink.
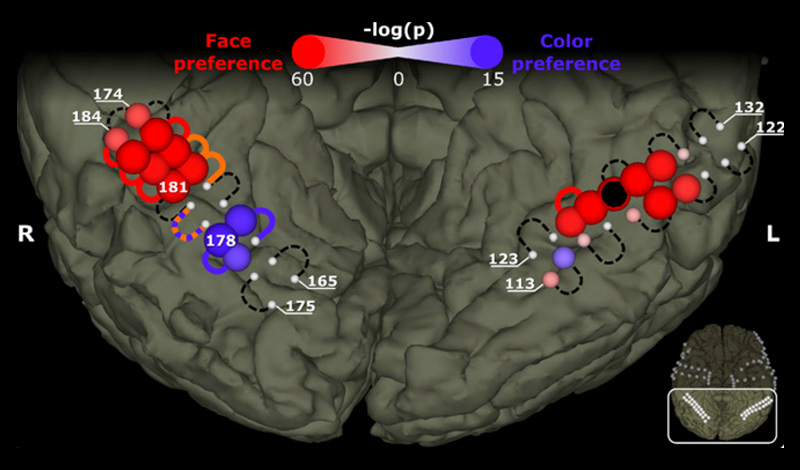
Many studies investigated the role of individual brain regions during dedicated mental processes to try to determine whether a specific brain region is exclusively engaged in a specific process. A recent study addressed that question by electrically stimulating the ventral temporal brain of a patient who underwent surgical treatment for epilepsy. Electrical stimulation of color- and face-selective sites, which were identified by means of visual evoked responses in the brain waves, affected only color and face percepts, respectively.
The patient reported effects such as illusory faces on top of objects and distorted perception of faces during stimulation of face-selective regions, and the appearance of illusory rainbows near color-preferring sites. These findings support the theory that the brain at least partly consists of specialized regions that are exclusively and causally engaged in a dedicated, specific mental process.
Intracortical brain stimulation is often performed in patients with epilepsy or tumor to identify the eloquent cortex. Doctors may stimulate motor regions that are responsible for movements of the finger, arm, leg, or other areas, and then observe the resulting movement. Doctors can also stimulate sensory regions to induce sensation and ask what the patient experienced. To map brain areas responsible for language, patients may be asked to name pictures while doctors stimulate the auditory cortex. If this stimulation interferes with the naming task, doctors have a better idea which regions produce language.
Median nerve stimulation may be performed by putting stimulating electrodes onto the arm close to the median nerve. This produces a hand movement and a somatosensory evoked potential (SEP) in the brain, which is measured with an ECoG area over the corresponding area. The central sulcus can be identified by detecting a phase reversal of the SEP in the recorded signal, which is an important landmark for neurosurgery.
Invasive stimulation of the cortex, and non-invasive nerve and muscle stimulation, have all been very helpful in our understanding of brain function. By stimulating the periphery, cortical responses can be produced, whereas stimulating the brain can produce motor movements. Both mapping strategies help us understand better cortical functions and perform brain-mapping experiments with a closed-loop system.
Treatment for patients suffering Essential Tremor or Parkinson’s Disease can include a Deep-Brain Stimulation (DBS) device. These devices emit short electrical pulses to directly stimulating brain tissue. In this approach, depth electrodes are implanted into the brain using a neuro-navigation system. The coordinates are extracted based on each patient’s anatomy. The exact positioning of the stimulation electrodes is essential for effectively helping the patient. The DBS control device is usually implanted in the patient’s chest, and is connected to the stimulation electrode mechanism in the brain. The effects of Essential Tremor or Parkinson’s Disease can then be attenuated by the systematic stimulation of the relevant brain area. This can dramatically improve patients’ quality of life by enabling them to move more freely through their daily activities. Every year, thousands of surgeries like this are performed worldwide.
The approach also bears some disadvantages. The electrodes are inserted into the brain until a certain position is reached, and the effects of the disease attenuate. However, this procedure occurs without consideration of relevant functional brain regions, and is therefore rather approximate. Next, the stimulation is performed steadily, and thus the brain is continually affected until the stimulator has to be exchanged when its battery is empty.
g.tec developed a research system that allows doctors to position the stimulation electrode based on precise knowledge of functional brain region and optimize the electrical stimulation to provide more flexible stimulation on demand. The optimization of stimulation parameters and timing reduced battery use, and thus the need for battery replacement. This procedure also paves the way for scientific studies for the optimization of DBS.
g.tec’s biosignal amplifiers and electrical stimulators can be used for setting up closed-loop systems to optimize DBS. Real-time analysis of biosignal data (local field potentials and/or multi-/single- unit activity) acquired from a variety of brain regions can be done to control the stimulation. It’s also possible to analyze the brain activity in real-time and stimulate different electrode positions for performance optimization. This would lead to better positioning and control of DBS electrodes and nearby brain areas, which in turn could further reduce tremor and other effects of Parkinson’s Disease, reduce battery power consumption via event-related stimulation, and enable rapid prototyping for scientific research in DBS.
Get Biosignal Amplifier
Get Cortical Stimulator
Get Switching Unit
SENSE, SWITCH & STIMULATE

Send us your email so we can contact you as soon as possible.